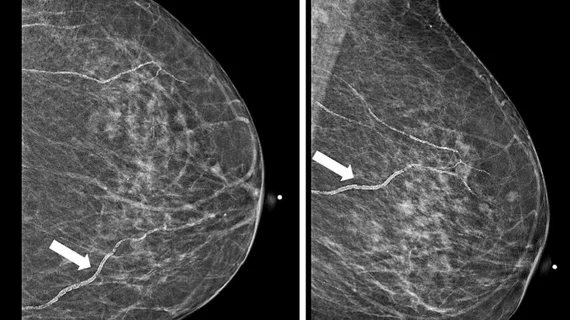
Wayne, NJ, JULY 27, 2016 – Konica Minolta announced today the release of its most advanced X-ray Digital Radiography (DR) system. Combining the unique built-from-the-ground-up design along with the fast and advanced Ultra image processing software, the system offers unprecedented workflow, clinical, patient convenience and satisfaction benefits. The new DR system (name not yet revealed) will be unveiled at the annual Association of Medical Imaging Management (AHRA 2016) conference in Nashville, July 31-August 3.